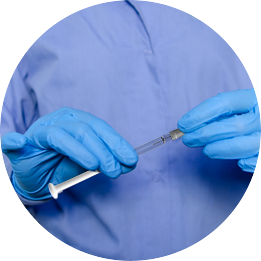
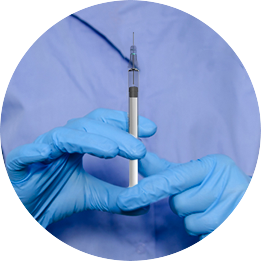
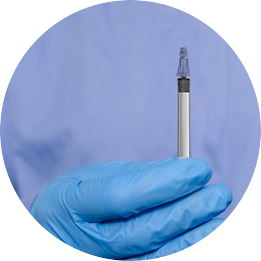
The Staked Companion allows conventional syringe procedures while providing end-of-dose cues, passive needlestick safety and reuse prevention

Pairing the right innovations creates a powerful synergy

The Staked Companion uses existing, commercially available primary package components




Protect your end users from needlestick and prevent reuse—the needle retracts into the plunger rod after use and is secured inside the barrel. Eliminate glue from your combination products, maintaining your drug integrity and enhancing patient safety.

Protect your end users from needlestick and prevent reuse—the needle retracts into the plunger rod after use and is secured inside the barrel.

The Companion is glue-free, removing the risk of unwanted interaction with the drug product and enabling alternative siliconization techniques.

Safeguard healthcare professionals and patients.